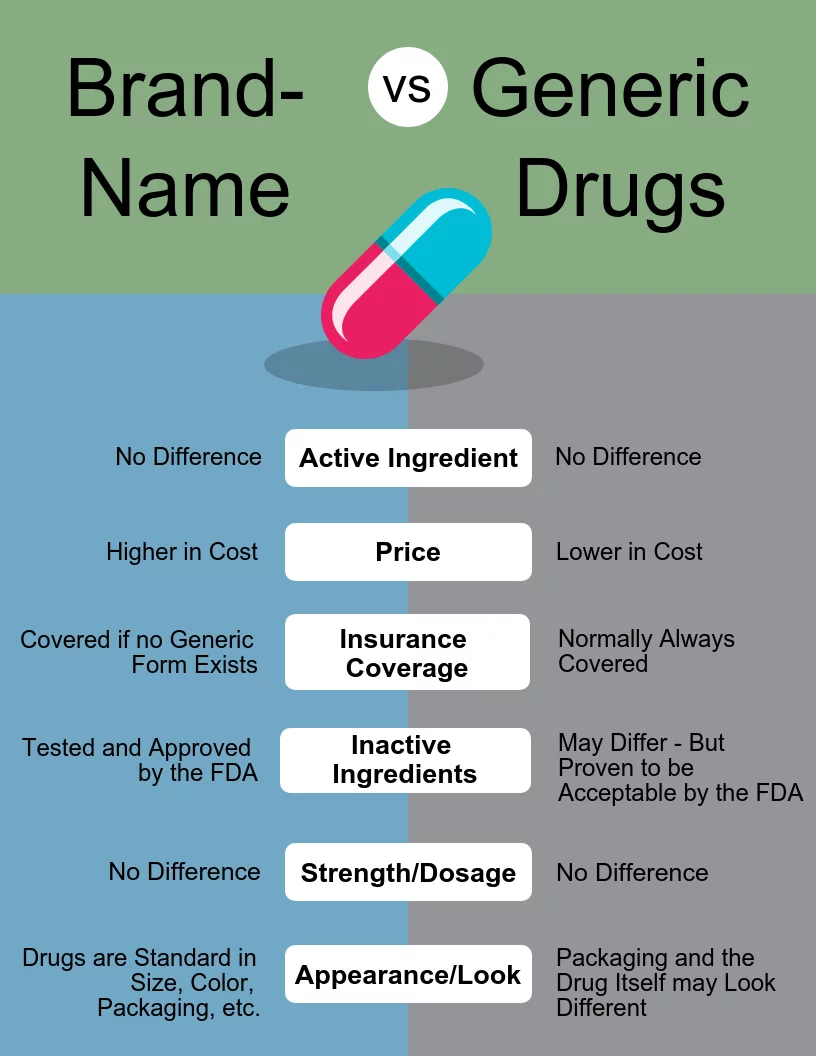
-
Drug AvailabilityInformation for consumers and health professionals on new drug warnings and other safety information, drug label changes, and shortages of medically necessary drug products.
-
Drug ShortageCurrent and resolved drug shortage/discontinuations reported to FDA.
On Shelf at CCBC Libraries
-
How Counterfeiters Are Contaminating America's Drug Supply by
Publication Date: 2005 -
Basic Pharmacology for Nurses by
Publication Date: 2017 -
Antibiotics Simplified by
Publication Date: 2018 -
Drugs in Society by
Publication Date: 2017
-
Medication GuideMedication guides are FDA approved documents that address issues that are specific to particular drugs, and can help patients avoid serious adverse events and side effects.
-
FDA AbbreviationA quick reference to acronyms & abbreviations related to FDA activities
Online from CCBC Libraries
-
FDA in the Twenty-First Century by
Publication Date: 2015 -
The Prescription Drug Abuse Epidemic by
Publication Date: 2018 -
Securing the Pharmaceutical Supply Chain by
Publication Date: 2015 -
A Bench to Bedside Approach by
Publication Date: 2012